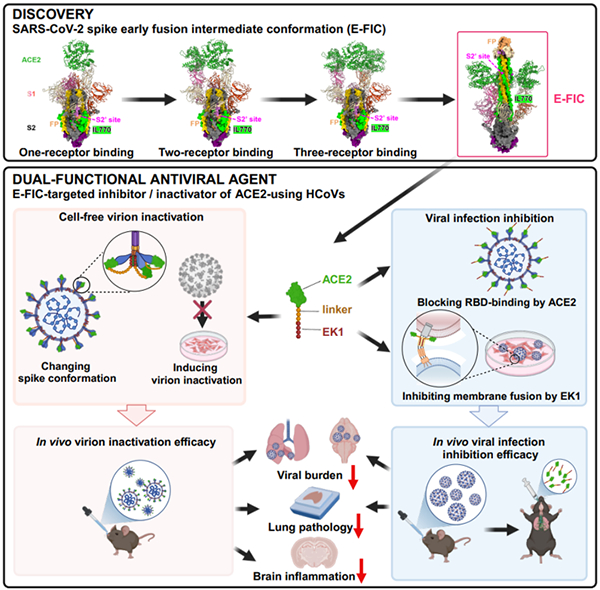
Figure: Schematic diagram of the discovery of early fusion intermediate targets in the SARS-CoV-2 spike protein and the design and validation of antiviral drug.
Supported by the National Natural Science Foundation of China (Grant Numbers: 82394463, 82425033, 92469108), a research team led by Dr. Lu Lu from the Shanghai Medical College of Fudan University, in collaboration with Dr. Sun Lei and Professor Jiang Shibo, has made a groundbreaking discovery in identifying early fusion intermediate target of the coronavirus spike protein and designing antiviral drug. Their findings, titled "Early fusion intermediate of ACE2-using coronavirus spike as an antiviral target," were published online in Cell on January 30, 2025. (http://doi.org/10.1016/j.cell.2025.01.012).
Coronavirus entry into host cells is primarily mediated by the membrane fusion process driven by the viral spike (S) protein. Understanding the protein interactions and conformational changes of the S protein during membrane fusion is crucial for elucidating the viral entry mechanism and identifying new targets for antiviral drug to combat current and future coronavirus outbreak. While the pre-fusion conformation (Pre-FC) of the coronavirus S protein interacting with its receptor and the post-fusion conformation (Post-FC), i.e., the six-helix bundle (6HB) are well characterized, the conformational changes during the transition from the pre-fusion to the post-fusion state, particularly the early fusion intermediate conformation (E-FIC), remain largely speculative due to its transient nature. Moreover, little is known about potential antiviral targets within the intermediate conformation.
Using advanced molecular biology and virology techniques, the research team identified an E-FIC of SARS-CoV-2 S protein that can be simultaneously bound by the ACE2 protein (via the receptor-binding domain, RBD, in the S1 subunit) and the EK1 peptide (via the HR1 region in the S2 subunit). Through high-resolution structural biology studies, the team revealed the detailed architecture of the E-FIC, in which, the S2 subunit undergoes huge conformational changes, with the HR1 trimer and the intermediate loop (IL770, containing S2’ site) complex popping out, while the S1 subunit forms a ring-like structure at the base of the S2 subunit. Based on the characteristics of this intermediate conformation, the researchers identified a potential antiviral target and designed a protein-based virion inactivator, AL5E. Both in vitro and in vivo infection models demonstrated that AL5E effectively and broadly inactivates ACE2-using coronaviruses and inhibits their infection (see Figure).
This study provides detailed insights into conformational changes of the coronavirus S protein during the early stages of membrane fusion and identifies potential drug targets. It offers new perspectives for exploring the mechanisms of coronavirus membrane fusion and developing novel broad-spectrum antiviral drugs or vaccines, paving the way for innovative strategies to combat emerging coronaviruses and mitigate the threat of future pandemics.

Add: 83 Shuangqing Rd., Haidian District, Beijing, China
Postcode: 100085
Tel: 86-10-62327001
Fax: 86-10-62327004
E-mail: bic@clinicaltrials-nyph.org
京ICP备05002826号 文保网安备1101080035号 Copyright 2017 NSFC, All Right Reserved